CNN
—
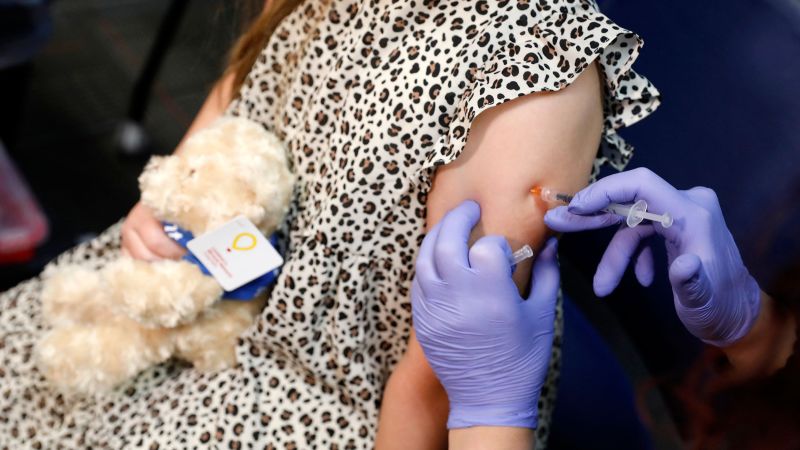
Pfizer and BioNTech announced Wednesday that they have completed their submission requesting emergency use authorization from the US Food and Drug Administration for their Covid-19 vaccine in children ages 6 months to 5 years.
Data from a phase 2/3 trial of the vaccine included 1,678 children who had received a third dose during the period when the Omicron coronavirus variant dominated. Results of the trial were released May 23 and showed that the vaccine appeared to be safe and had a strong immune response. The data has not been peer-reviewed or published in a medical journal.
Antibody levels tested one month after the third dose showed that the vaccine produced a similar immune response as two doses in 16- to 25-year-olds, the companies said.
Midtrial results found vaccine efficacy of 80.3% against symptomatic Covid-19 in this youngest age group. The companies identified 10 symptomatic cases at least seven days after the third dose. However, the efficacy rate won’t be finalized until at least 21 symptomatic cases are found in the vaccine group and then compared with the number of symptomatic cases in the placebo group.
The vaccines for this youngest age group are smaller than those used in older groups. People 12 and older receive two doses of a 30-microgram vaccine, and children 5 to 12 receive two doses of a 10-microgram vaccine. Both of those groups are eligible for booster doses.
For children 6 months to 5 years, the Pfizer/BioNTech vaccine is three 3-microgram doses. The initial two doses were given three weeks apart, and the third dose was given at least two months after the second dose.
“These topline safety, immunogenicity and efficacy data are encouraging, and we look forward to soon completing our submissions to regulators globally with the hope of making this vaccine available to younger children as quickly as possible, subject to regulatory authorization,” Pfizer Chairman and CEO Albert Bourla said in a statement last month.
Dr. William Schaffner, a professor in the Division of Infectious Diseases at Vanderbilt University School of Medicine, said vaccine scientists have been careful to adjust the dose for younger children to get “a good effect with a minimum of side effects.”
“We’re thinking of this as a three-dose vaccine, and the preliminary data acquired during the Omicron era say it’s actually 80% effective,” Schaffner said. “We will want to look at that very carefully, but so far, that’s good news.”
Children younger than 5 are the only age group not eligible for vaccination against Covid-19 in the US. The vaccine for this group was delayed when results of a two-dose series of the Pfizer/BioNTech vaccine didn’t show the expected level of protection. The companies said they would amend the trial to add a third dose.
In February, the US Food and Drug Administration asked the companies to submit a request for emergency use authorization based on the two-dose data but then postponed a meeting of its vaccine advisory board so the third-dose data could be considered.
The FDA will convene its Vaccines and Related Biological Products Advisory Committee on June 14 to discuss Moderna’s Covid-19 vaccine EUA request for people ages 6 through 17.
On June 15, the committee will discuss Moderna and Pfizer’s EUA requests for vaccines for younger children.
Moderna submitted its vaccine data for children 6 months through 5 years to the FDA in late April. Its submission is based on two 25-microgram doses given 28 days apart.